-

1992
The Institute of Biomedical Engineering, Chinese Academy of Medical Sciences pioneered the development of ophthalmic ultrasound in China.

-

1992
The Institute developed the first Ultrasonic A/B Scanner for Ophthalmology in China.
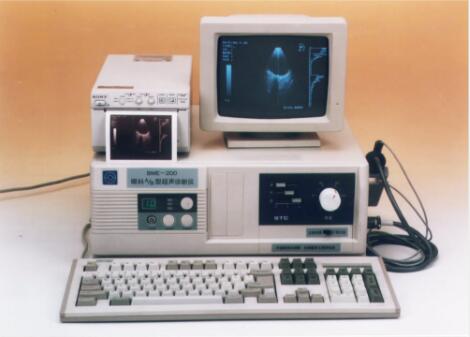
-

2000
The Institute developed the first Ultrasound Biomicroscope in China.
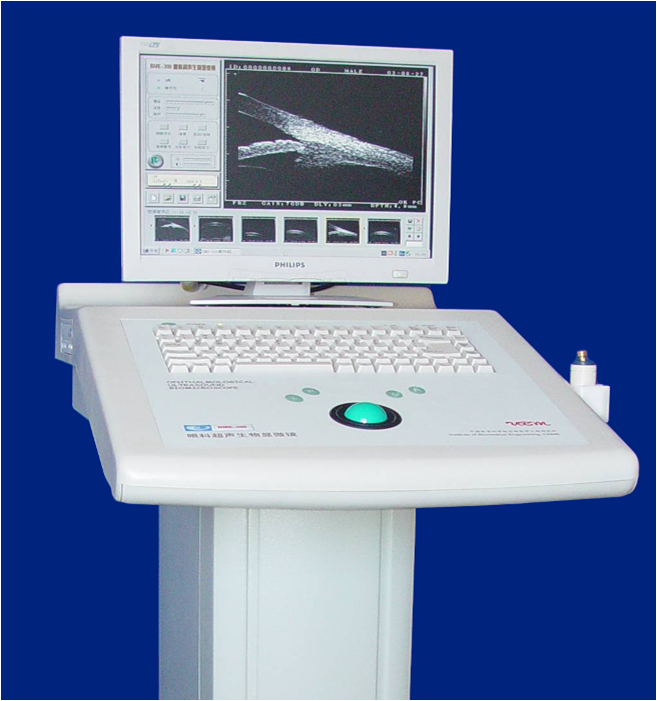
-

2004
MEDA was co-founded by the backbones of the Institute.

-

2005
MEDA launched its Ultrasonic Biometer and Pachymeter.

-
2005
MEDA obtained its first CE and ISO certificates, establishing its presence in European markets.
-

2007
MEDA obtained its first FDA clearance, ensuring its entry into U.S. market.
-

2009
MEDA was successfully acquired by Tianjin Pharmaceutical Holdings Ltd.

-

2010
MEDA launched its new Ultrasonic Bladder Scanner
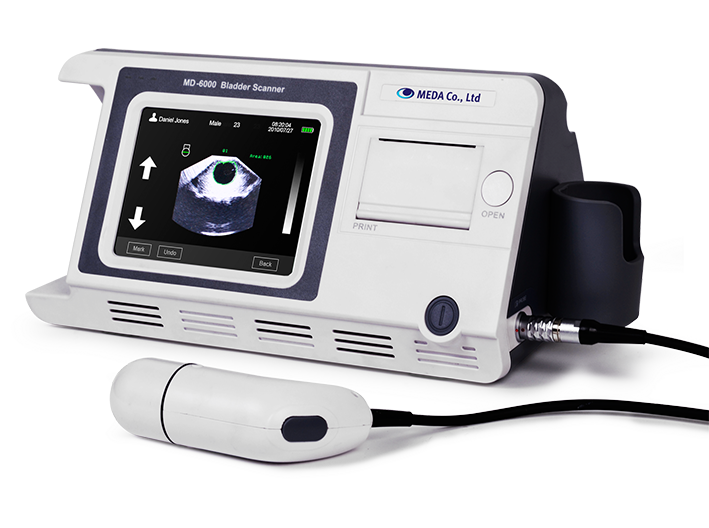
-

2012
MEDA launched MD-2300S, its flagship of Ultrasonic A/B Scanner for Ophthalmology.
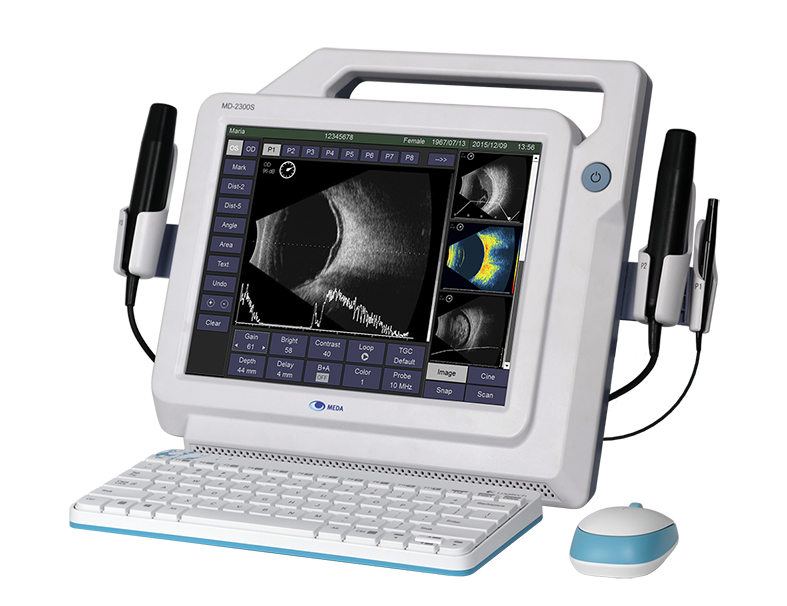
-

2012
MEDA launched its Nd:YAG Laser for Ophthalmology.

-

2013
MEDA went public on NEEQ (430220).

-

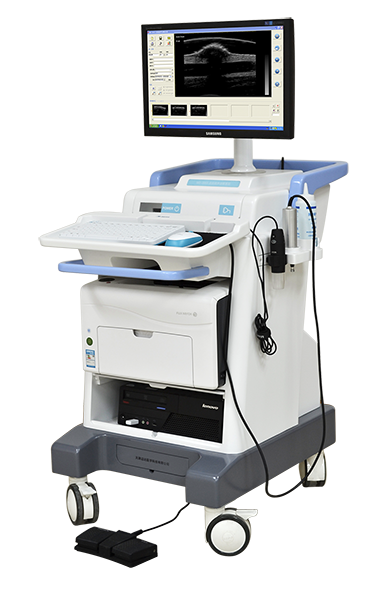
2014
MEDA developed the first Ultrasonic Skin Scanner in China.
-

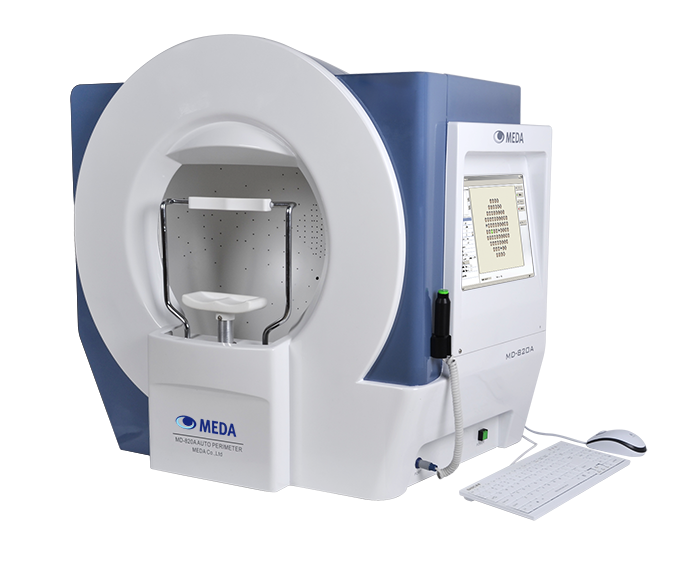
2014
MEDA launched its new Auto Perimeter.
-

2014
MEDA developed the first Nd:GdVO4 Photocoagulator in China.

-

2018
MEDA launched its Wireless Ultrasonic Bladder Scanner.

-
2018
MEDA obtained NRTL certification.
-

2018
MEDA introduced a line of fundus camera products to complement its ophthalmic solution.

-

2019
MEDA celebrates its 15th anniversary, and will stay focused on innovation and quality commitment.

-
PRODUCTS
- Biometer
- Pachymeter
- Phaco
- Perimeter
- Biometer
- Pachymeter
- Phaco
- Perimeter
- Biometer
- Pachymeter
- Phaco
- Perimeter
- Biometer
- Pachymeter
- Phaco
- Perimeter
- Biometer
- Pachymeter
- Phaco
- Perimeter
- Skin Scanner
- Middle-Ear Analyzer
- EVENTS
- SUPPORT
+
Follow us on Facebook
+
Follow us on WeChat
 Milestones
MilestonesTel: 0086-22-83713828
Fax: 0086-22-83713880
Email: export@meda.com.cn
Contact Us | Legal | Site Map
© 2024 MEDA Co., Ltd. Tianjin, China All rights reserved Support by kbyun.com
